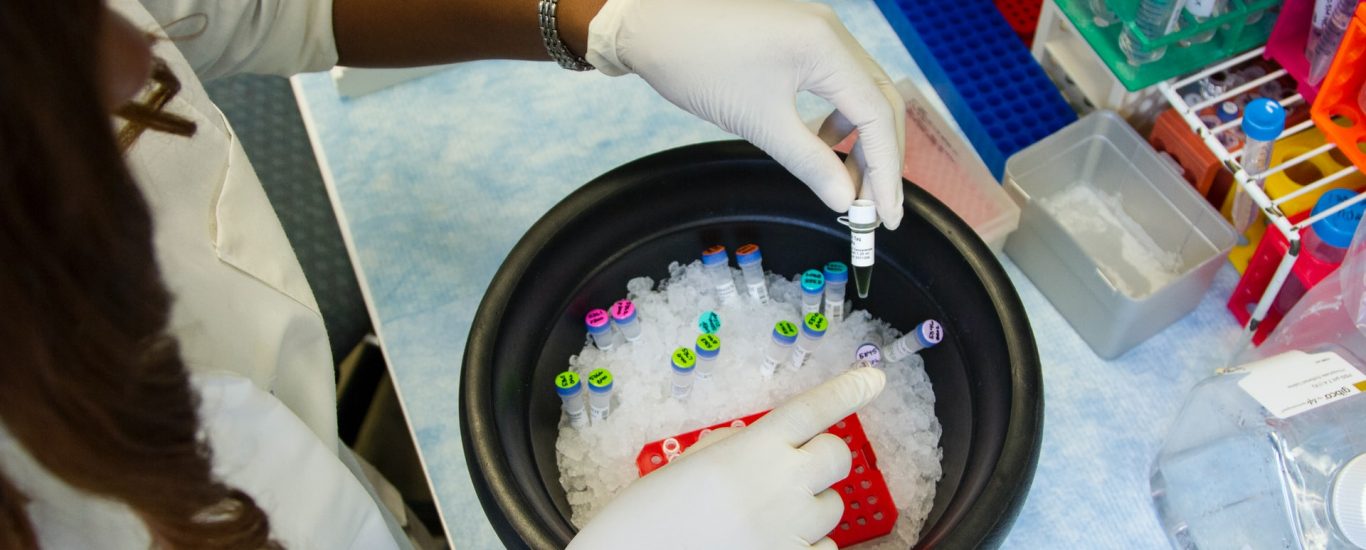
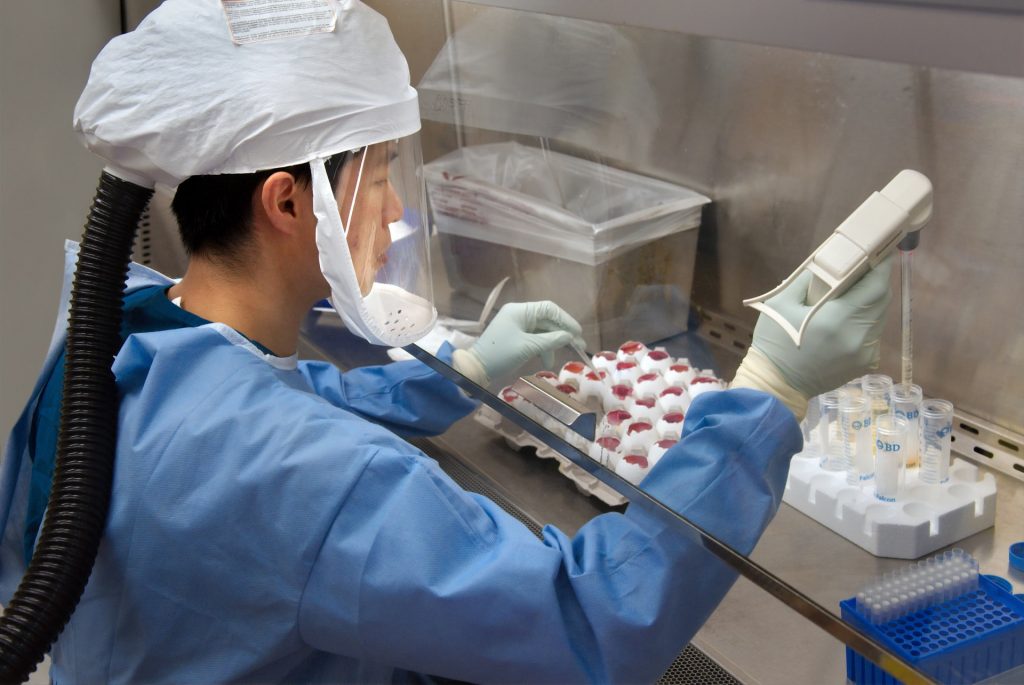
Validation of clinical study is an important part of every trial, and it determines if a medical study can be considered valid. Researches have been working hard to create a vaccine that can tackle covid-19, and for now, they have succeeded. A vaccine has been released and has been proven to be effective. For the vaccine to work accurately, then there would have been many different elements that had to be checked and validated throughout the process in order to achieve the accurate results at the end. Since clinical trials are extremely important in the way in which we find new cures for diseases, each part of the trial must be taken seriously and must comply with all the rules and regulations. Validation of clinical study at each stage will ensure that the information passed on to the next stage is in fact reliable and 100% accurate to get passed on to the next stage.

Validation of Clinical Study Prevents Errors
One reason why validation of clinical study is so important is that clinical research involves a lot of statistical analysis, and this has to be done by qualified individuals. When there are errors made in these statistical calculations, then the conclusions that a researcher draws about the benefits and risks of a given drug may not be entirely accurate. A mistake in the calculations can cost someone their life or severely limit their ability to continue living a normal life. This is the reason why validation is so important for clinical trials. Without validation of clinical study, there is no way to determine whether the results of the study have been accurate or not.

Why Do We Need Clinical Trials?
Clinical trials are absolutely necessary to the development of effective new treatments which help individuals to live better and with less disability or illness. With this in mind, it is very important to have as much information as possible about trials, and the validation of clinical study stages becomes clear. Each validation stage helps to make sure that the data about every treatment that was tested has been collected correctly. Without clinical trials, new medications could not become available to individuals suffering from diseases that have no cure at present. However, there are also many pharmaceutical companies that spend millions each year conducting clinical trials in order to prove their drugs work. The reason why clinical trials need to be conducted correctly is that otherwise, it would be impossible for them to receive any financial incentive for the trials if they prove to be unsuccessful. For example, if a drug is discovered to be highly effective and then proves to be unsafe, there would no way for the company to receive compensation for the loss incurred. If the drug proves to be ineffective, they could end up losing millions in research funding that they spent to make the drug.

James is a furniture designer specializing in ergonomic and stylish custom office desks. He focuses on blending functionality with modern aesthetics to create workspaces that enhance productivity.